
Cannabis Shelf Life: What Data Tells Us
Shelf life is often left to regulators who don't often have background in cannabis science. What data can we use to help further the discussion around cannabis and shelf life?
Stay in touch!
Join our email list to receive Calyx content & product updates.
By Tobe Nightengale in Cannabis Info , Cannabis Packaging
Quantifying Shelf Life for Cannabis Products
Cannabis is a complex plant, containing over 150 Terpenes, 100 unique Cannabinoids, and still many other Volatile and Cannflavin compounds. Each of these Cannabis Constituents offer individual, and entourage, benefits for the consumer, but how can we determine how fresh your product really is?
Many methods for quantifying shelf-life have been explored : Moisture Content, THC or CBD Concentration, Terpene Concentration, Microbial Contamination, to name a few.
The lack of a universally accepted method for establishing the rate of Cannabis degradation makes it extremely difficult for producers and retailers to know how long they have to sell product, and what conditions to store it in until final sale. Ambiguity in Cannabis Shelf Life poses a particular concern for medical consumers, who often purchase products due to the Cannabinoid and Terpene Profile at the time of initial testing. However, these characteristics can change drastically in just a few short weeks of storage.
What Does Cannabis Degradation Mean?
Spoilage, degradation, decomposition, and contamination are familiar terms in nearly every industry that packages an organic product that will, eventually, no longer be fit for consumption.
Most people understand the importance of checking expiration dates on food, medicine, and cosmetics to make sure that the quality of the product is the same (or similar) to the quality at the time of packaging. Labels such as “use before”, “sell by”, “expires on” all give indication to the amount of time a product can stay in its original packaging before it is no longer at optimal quality. However, even these terms carry a degree of obscurity, and the FDA has become more outspoken that many of these labels are not determined by an exact science.
To date, there is no accurate shelf-life associated with cannabis material, and many states will regulate stored cannabis products with an arbitrary date.
This results in many products being discarded too early (or too late…), contributing to a massive waste and environmental impact driven by the cannabis industry. While there are certainly some characteristics that make a cannabis product not fit for sale (such as heavy metals, coliforms, pesticides, etc.), many secondary degradation products result in an effect change, rather than a spoilage.
In order to explore the shelf-life of cannabis, a more complex approach must be taken. At its most plain, the process of degradation is characterized as a reduction in the complexity of a chemical compound. Heat, light, and air constantly work together to force complex molecules into a simpler (entropically favorable) structure. For cannabis, this means the transition of “desirable” compounds into “undesirable” compounds. One prime example of this is the degradation of Δ9-THC into CBN:
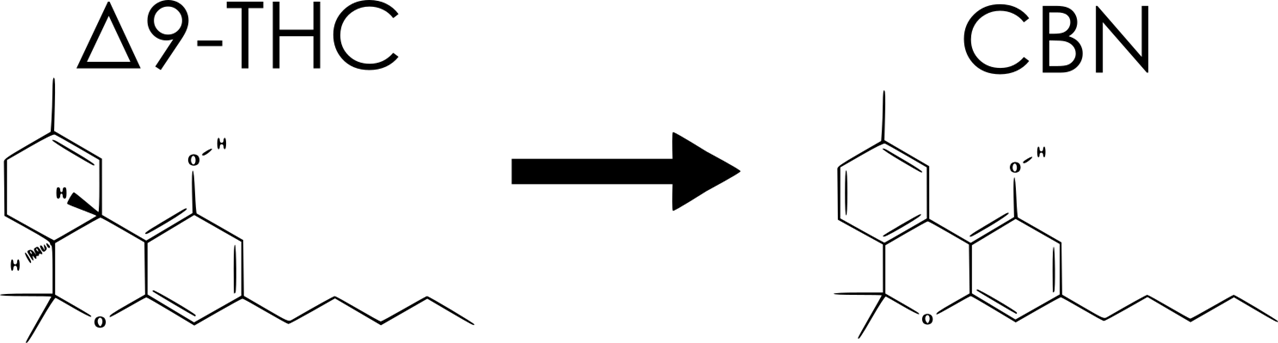
If you’ve ever consumed a cannabis product that has sat around on a sunny windowsill, or tucked away in a drawer for a year or two, you may have noticed that it had a significant drowsy effect. This is directly caused by CBN in tandem with Δ9-THC. This happens to be one of the first “entourage effects” discovered, and is of primary concern when it comes to storing cannabis. Drowsiness is generally considered to be an undesirable effect, but an effect nonetheless, so this can’t possibly be the criteria for shelf-life, right?
As a matter of fact, cannabis products with high levels of CBN are gaining popularity as a Sleep Inducer for those who cannot sleep regularly. So if a cannabis product tests high in CBN concentration, is it considered waste to be discarded, or considered a new product to be re-labelled and sold?
This is just one example of the difficulties in determining accurate shelf-life of cannabis products, especially since testing is only performed upon initial product packaging. Multi-phase testing at key timepoints is one way to obtain a better understanding of where a product is in its degradation process, as opposed to when a product has reached its expiration date based on initial test results.
How Does Calyx Quantify Cannabis Degradation & Shelf-Life?
Since no single method of analysis is reliable in the current industry, we take a broad approach to understand what compounds are degrading, the rate at which they degrade, and the subsequent concentration of secondary and undesirable degradation products. To correctly perform Shelf-Life Testing, we look at a wide variety of Major and Minor Cannabinoids, as well as Terpene Concentration and Safety Testing.
Cannabis Analyte Overview
TAC
TAC, or Total Active Cannabinoids, is a measure of the sum of all Major and Minor Cannabinoids present in a sample. This test target offers a high-level, if imprecise, view of what types of degradation are occurring.
While it would seem that TAC should always decrease over time, the reaction is highly dependent on the characteristics of the Cannabis product being tested. Full spectrum products which have been exposed to minimal heat and contain a raw Cannabinoid + Terpene Profile may experience an increase in TAC due to the continual synthesis of new Cannabinoids from Cannabinoid pre-cursors (such as CBG-A). In products with very low concentrations of Cannabinoid pre-cursors, we can expect the rate of degradation to exceed the rate of natural synthesis.
On the other hand, distilled products, or products that have experienced greater heat or filtration, are less likely to experience natural Cannabinoid synthesis, and will overwhelmingly exhibit a decrease in TAC over time.

THC-A
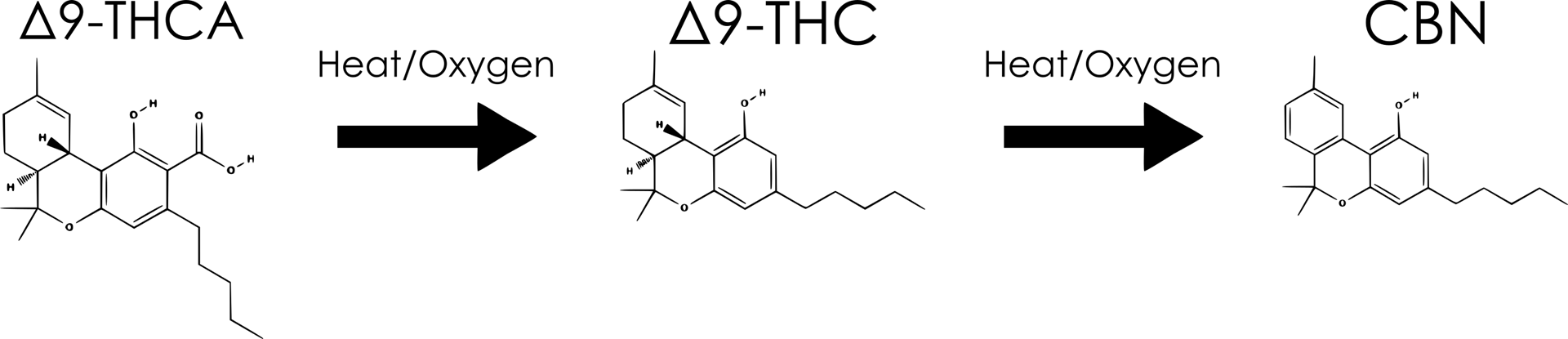
The concentration of THC-A is an important marker to determine the rate of degradation of a Cannabis product. This is a compound that most producers are familiar with, but is often confused or combined with Δ9-THC at the consumer level. Similar to TAC, this concentration can increase or decrease with time, depending on the characteristics of the Cannabis product. THC-A concentration can change by two different reaction pathways : Synthesis and Decarboxylation.
When conditions are favorable, CBG-A will naturally synthesize new THC-A, even in a packaging environment! This is a mostly beneficial reaction, with increasing THC-A allowing for greater overall potency of the final product, even after long-term storage.
However, the secondary decarboxylation of THC-A indicates a potential for less desirable outcomes. While decarboxylation is typically known to be a heat-mediated reaction, it will also occur in room temp and cold temperature environments as a spontaneous reaction. Oxygen is the true catalyst for decarboxylation, so minimizing Oxygen Transfer in a packaging environment is key to maintaining (and potentially increasing!) THC-A concentration after packaging.
Δ9-THC
The concentration of THC, and subsequent formation of CBN, is an ideal indicator for the overall quality of a packaged Cannabis product. The relative simplicity of this reaction pathway allows for accurate insights into the product’s exposure to heat and oxygen. Since this is a balanced, two-step reaction, a decrease in THC inevitably results in an increase in CBN, an undesirable degradation product.
Both of these reaction steps are mediated by Oxygen exposure, so high rates of change in these compounds indicate a high rate of Oxygen Transmission in the packaging.
CBN
A one-step process, the isomerization of THC results in the single degradation product of CBN. Under normal storage conditions, THC will almost exclusively degrade into CBN, making it another ideal target for investigation.
Increases in CBN concentration over time indicates high rates of heat and oxygen exposure within the packaging.
While this can be an ideal target, the extremely low starting concentrations of CBN, as well as the relatively small changes due to degradation over shorter time periods, can make it difficult to track accurately. Analytical methods outside of High Pressure Liquid Chromatography / Mass Spectrometer will often have a large degree of variance when tracking the concentration of this compound, so it is recommended to pull insights from a combination of THC and CBN degradation data.
CBD-A
The concentration of CBD-A is another indicator that good storage conditions are being met, and that obtained shelf life data is accurate. While not a particularly good test target for tracking degradation, CBD-A concentration can act as a Positive Control, where Cannabis material containing CBG-A should exhibit a small, but consistent, increase in CBD-A over time.
If CBD-A concentration is falling or staying the same under normal storage conditions, the confidence in other obtained analytical data is low.
CBD
Dissimilar to the THC synthesis pathway, the decarboxylation of CBD-A into CBD requires a significantly larger energy input, and will overwhelmingly occur as a result of direct heat exposure.
In normal storage conditions, the increase in CBD concentration should be negligible. In this way, CBD concentration can effectively act as a Negative Control, where no identified rate of change indicates confidence in other obtained data.
If CBD concentration is increasing or decreasing at a significant rate, it can mean that obtained analytical data is inaccurate, or that proper storage conditions are not being met for the testing period.
Terpenes
While the evidence for significant consumption and / or medical benefit is currently limited, Terpene concentration and retention is of high concern for many producers and consumers.
Terpene Retention poses the greatest challenge to the proper packaging and storage of cannabis products due to their inherent volatility and solvent properties. Terpene retention is only one factor which dictates the quality of packaged Cannabis products, and must be balanced with the retention of other key Cannabis constituents.
Additionally, unlike other Cannabis compounds, the degradation of Terpenes by Oxygen, Heat, and UV Light is negligible, and preservation of these compounds is almost entirely dependent on seal quality and container component materials.
Conclusion
The complex nature of cannabis with its diverse constituents, including terpenes, cannabinoids, and volatile compounds, poses a significant challenge in determining its shelf life. Various methods, from measuring moisture content to terpene concentration, have been explored to quantify degradation, yet a universally accepted approach remains elusive. This lack of clarity impacts producers, retailers, and particularly medical consumers seeking consistent cannabinoid and terpene profiles. The absence of well studied and quantified data for regulators and producers leads to premature waste and environmental concerns.
Request a sample kit to test our products for yourself.



